Lattice Energy Trend Lattice Energy Examples Hence, the lattice energy increases from left to right across a period and decreases from top to bottom down a group. 
Across a period, the atomic charge increases, and down a group, the ionic radius increases. It is clear that the bond between Na + and OH – (NaOH) has the smallest lattice energy, and that between Al 3+ and O 2- (Al 2O 3) has the greatest.įrom the above tables, one can observe that the lattice energy increases with atomic charge and decreases with ionic radius. The following table shows the lattice energies for salts of OH – and O 2. The lattice energy is proportional to the product of the two ionic charges ( ΔH lattice∝ | Q 1Q 2|). In other words, the ionic bond becomes stronger as the charge on the ions becomes large. 
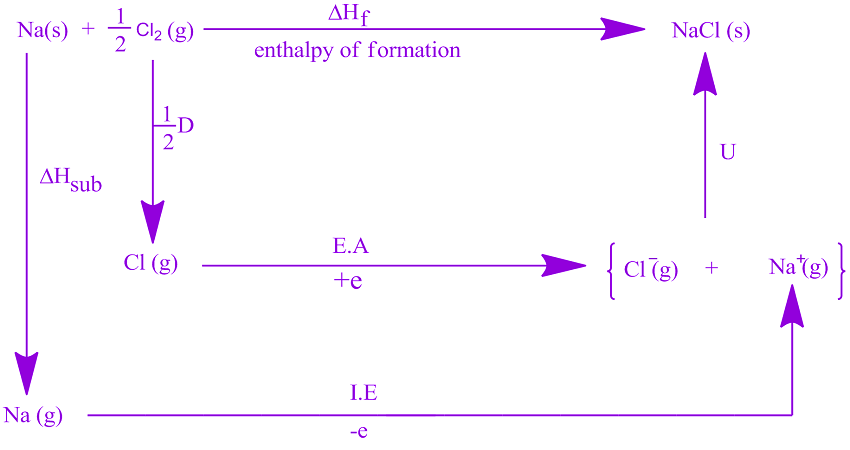
Atomic charge: As the atomic charge increases, the lattice energy increases. It is clear that the bond between Li + and F – (LiF) has the highest lattice energy and that between Cs + and I – (CsI) has the lowest.Ģ. The following table shows the lattice energy values (in kJ/mol) for the ionic bond formed between alkali metals and halogens. In other words, the bond between opposite ions is strongest when the ions are small. Ionic radius: As the ionic radius increases, the lattice energy decreases. Lattice energy can be calculated using electrostatics or estimated from the Born-Haber cycle. The lattice energy magnitude of an ionic crystal can be determined from the following equation derived from Coulomb’s law.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
